Safety of using IUDs and PEMFs
Safety of Using IUDs and PEMFS Table of Contents Long-acting reversible contraceptives (LARCs), such as intrauterine devices (IUDs) are in common use for pregnancy prevention.
This section of our website will profile short reviews of important recent PEMF studies. People often ask me for scientific proof supporting PEMFs. While creating new content, we continuously find interesting and compelling research on PEMFs you will not likely find elsewhere and which you should find valuable.
Check back regularly for new information.

Safety of Using IUDs and PEMFS Table of Contents Long-acting reversible contraceptives (LARCs), such as intrauterine devices (IUDs) are in common use for pregnancy prevention.

Use of a PEMF to Treat Complex TBI with Brain Gauge and Rivermead Outcome Measures Table of Contents Journal of Science and Medicine Abstract by

The Role of Pulsed Magnetic Fields (PEMFs) in the Management of Concusion and Traumatic Brain Injury (TBI) Table of Contents Journal of Science and Medicine
Age-Associated Memory Impairment Reversed with PEMF Therapy Table of Contents About 40% of people over age 65 experience some memory loss. It is known as
Diabetic Foot Ulcer Infections – Combining Therapy with PEMF and Lasers Table of Contents I have often said that combining PEMF therapies with other modalities

Essential Tremor and Treatment with PEMF Devices Table of Contents ESSENTIAL TREMOR AND TREATMENT WITH PEMF DEVICES Essential tremor (ET) is a neurological condition that
PEMFs Reverse the Muscle Damage Caused By Cholesterol Lowering Drugs (Statins) Table of Contents STATIN DRUG USE One in four Americans, ages 45 and older
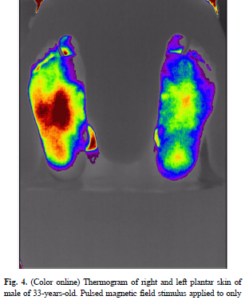
PEMF for Microcirculation and Increasing Blood Flow Table of Contents CAN PEMFS HELP IMPROVE BLOOD FLOW AND CIRCULATION? I have previously stated that almost all
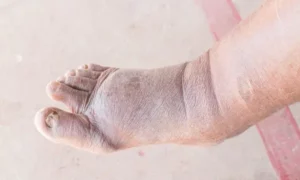
PEMFs for Healing Diabetic Foot Ulcers Table of Contents Diabetes is an increasing problem around the world. Vascular disease is one of the most common
PEMFs Reduce Progression of Arteriosclerosis Table of Contents Vascular disease within the heart and throughout the body is one of the top causes of death
Pulsed ElectroMagnetic Fields Help with Muscle Soreness After Exercise Table of Contents MUSCLE SORENESS FROM EXERCISE Delayed onset muscle soreness [DOMS] is a common painful
Urinary Stress Incontinence Improved By High Intensity PEMF Therapy Table of Contents WHAT IS STRESS INCONTINENCE? Stress incontinence, primarily urinary, is a very common problem
PEMFS AND BRAIN RECOVERY AFTER STROKE Table of Contents The ability to recover after stroke depends on many factors, including the regenerative capabilities of the brain.