PEMFs and Parkinson's (PD)
Table of Contents
PEMF treatment of Parkinson patient – Case Report
A 74-year-old retired building inspector with a 15-year history of Parkinson’s disease (PD) presented with a severe resting tremor in the right hand, generalized bradykinesia, difficulties with the initiation of gait with freezing, mental depression, and generalized cognitive impairment despite being fully medicated. Testing of constructional abilities employing various drawing tasks demonstrated drawing impairment compatible with severe left hemispheric dysfunction. He had two successive transcranial treatments, 20 minutes duration each, with pulsed electromagnetic fields (PEMFs) of very low intensity and frequencies of 5Hz and 7Hz. His tremor disappeared, and he had dramatically improved drawing performance. As he continued daily treatments with PEMFs for two more days, he had additional striking improvements in his drawing. This treatment demonstrates rapid reversal of drawing impairment in Parkinson’s disease, related to left hemispheric brain dysfunction even by brief transcranial very, low-intensity PEMFs. It also shows that cognitive deficits associated with Parkinsonism, which usually are progressive and unaffected by dopamine replacement therapy, may be partly reversed by these PEMFs. Treatment with PEMFs reflects a “cutting edge” approach to the management of cognitive impairment in Parkinsonism. (Sandyk, Sep 1997)
Parkinson’s Disease Introduction
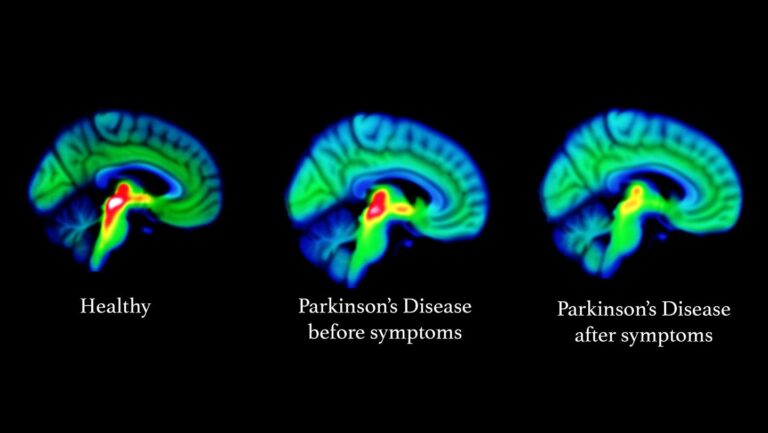
Parkinson’s disease (PD) is the second most common neurodegenerative disorder in people over the age of sixty. Aging is the major contributing factor for increased risk of developing Parkinson’s disease. With the aging of the population worldwide, the frequency of Parkinson’s disease is expected to increase dramatically in the coming decades. Nearly one million people in the US are living with Parkinson’s disease. It is estimated that 6-10 million people worldwide have Parkinson’s disease, affecting all races and ethnicities. The incidence of Parkinson’s disease rises rapidly with age, affecting approximately 1% of the population older than sixty years and approximately 4% of those older than eighty years. Every nine minutes, someone in the United States is diagnosed with Parkinson’s disease. Parkinson’s disease (PD) is a chronic and progressive movement disorder primarily, meaning that symptoms continue and worsen over time. The cause is unknown, and there is no cure.
Conventional treatment options include medication and surgery to manage symptoms. Neither reduces progression.
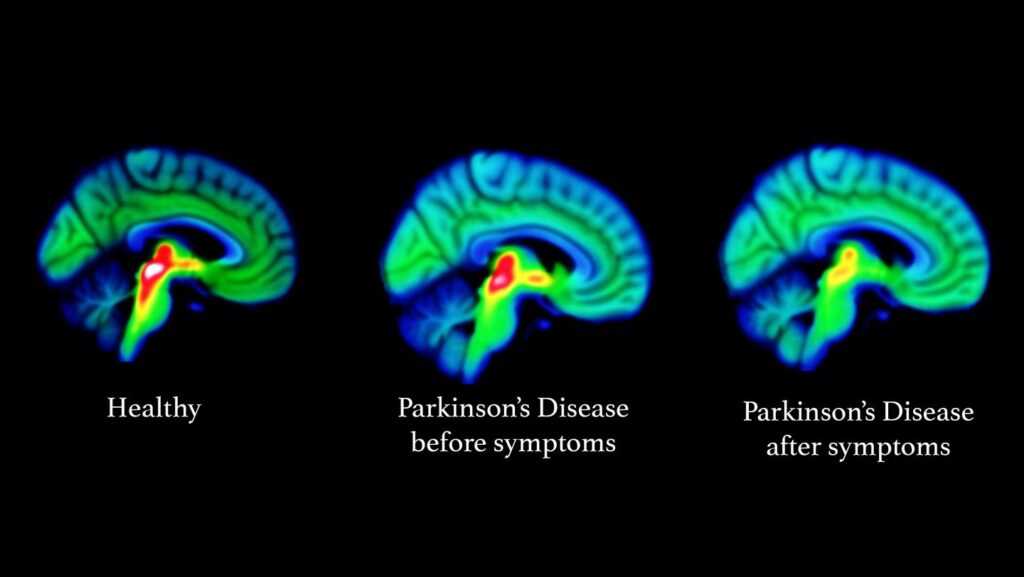
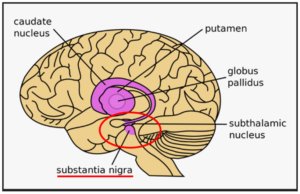
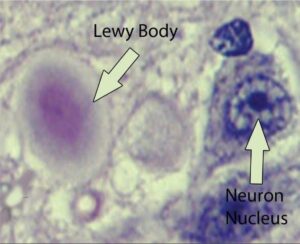 Parkinson’s disease most obviously involves the malfunction and death of vital nerve cells (neurons) in the brain, primarily an area of the brain called the substantia nigra. Some of these dying neurons produce dopamine, a neurochemical that sends messages to the part of the brain that controls movement and coordination. As Parkinson’s disease progresses, the amount of dopamine produced in the brain decreases, leaving a person unable to control movement normally. Loss of cells in other areas of the brain and body contribute to Parkinson’s. For example, researchers have discovered that the hallmark sign of Parkinson’s disease—clumps of a protein alpha-synuclein, which are also called Lewy Bodies—are found not only in the mid-brain but also in the brain stem and in scent cells. These areas of the brain correlate to non-motor functions such as sense of smell and sleep regulation. The presence of Lewy bodies in these areas could explain the non-motor symptoms experienced by some people with Parkinson’s disease before any movement (motor) signs of the disease appear. The intestines also have dopamine cells that degenerate in Parkinson’s, and this may be important in the gastrointestinal symptoms that are part of the disease.
Parkinson’s disease most obviously involves the malfunction and death of vital nerve cells (neurons) in the brain, primarily an area of the brain called the substantia nigra. Some of these dying neurons produce dopamine, a neurochemical that sends messages to the part of the brain that controls movement and coordination. As Parkinson’s disease progresses, the amount of dopamine produced in the brain decreases, leaving a person unable to control movement normally. Loss of cells in other areas of the brain and body contribute to Parkinson’s. For example, researchers have discovered that the hallmark sign of Parkinson’s disease—clumps of a protein alpha-synuclein, which are also called Lewy Bodies—are found not only in the mid-brain but also in the brain stem and in scent cells. These areas of the brain correlate to non-motor functions such as sense of smell and sleep regulation. The presence of Lewy bodies in these areas could explain the non-motor symptoms experienced by some people with Parkinson’s disease before any movement (motor) signs of the disease appear. The intestines also have dopamine cells that degenerate in Parkinson’s, and this may be important in the gastrointestinal symptoms that are part of the disease.
Standard medication treatment
Standard medication treatment of Parkinson’s disease can be only 50% effective overall. As the dose of taken medication is wearing off, there can be a decline in the drug’s efficacy. For example, in the morning shortly after taking it, the medication may be 90% effective, in the afternoon only 50% effective and in the evening only 30% effective. Twice-weekly treatments with extremely low intensity PEMFs applied to the head for ten weeks has been shown to eliminate these medication declining efficacy symptoms. At ten weeks after starting the PEMFs, there was 40% improvement in response to medication with minimum change in efficacy during the course of the day or evening. PEMFs appeared to enhance response to medication. Since decline in the response to medication is a phenomenon associated with progression of the disease, these results suggest that intermittent application of PEMFs may reverse the course of progressive Parkinson’s disease (Sandyk, Oct 1997).
Major aspects of Parkinson’s disease that are not addressed by conventional medical therapies, specifically pharmaceuticals, can be significantly impacted by PEMFs. These include: Neuro-inflammation, the Brain-Gut Axis, Ferroptosis, Mitochondrial Dysfunction, the Braak Hypothesis and Alpha-synuclein, Vagal Nerve Stimulation (VNS) and Deep Brain Stimulation (DBS), medication side effects, stem cell therapy, and comorbidities.
The major role of PEMFs in Parkinson’s disease is to impact multiple aspects of the condition, unlike most other therapies which tend to be one-dimensional. In any given individual with Parkinson’s disease there are often multiple factors that play into Parkinson’s disease, both in terms of initiation, progression and holistic management. PEMFs address multiple factors simultaneously because of their general actions in the body, covered in the books Power Tools for Health and Supercharge Your Health with PEMF Therapy and on the website DrPawluk.com. The impact of PEMFs on all of the above Parkinson’s disease contributing factors, are all covered in greater depth in the PEMFs and Parkinson’s Disease e-book. See the e-book section on Dr. Pawluk.com.
Pulsed Electromagnetic Fields (PEMFs) in Parkinson’s disease
The exploration of Pulsed Electromagnetic Fields (PEMFs) in Parkinson’s Disease (PD) represents an innovative frontier in neurodegenerative disorder treatment. The application of PEMFs offers a non-invasive method to potentially mitigate symptoms and alter the disease’s progression through mechanisms that influence cellular and neurological functions.
Electromagnetic stimulation of is a non-invasive rapidly emerging biological tissue treatment technique. PEMFs induce ion currents in the tissue and depolarize the electrical activity of all membranes, neurologic and non-neurologic slightly. Electromagnetic stimulation in animals and in the laboratory enhances cellular activity and stimulates growth-related responses and regeneration. As a result, PEMFs stimulate nerve growth and rebalance nerve abnormalities, increase microvascular blood flow and tissue oxygenation, and increase the amount and density of capillaries. As a result, PEMF treatment would be expected to delay disease progression and even induce neuro-repair in Parkinson’s disease. (Jensen)
Research on the use of PEMFs in Parkinson’s disease
PD Symptom | Comments | Ref |
Yawning and stretching | These are dopamine behaviors. Extremely low intensity PEMFs have been found to increase yawning and stretching. | Sandyk, Mar 1999 |
Smell | The smell center of the brain also contains large amounts of dopamine neurons. Anti-Parkinson’s drugs do not affect the smell threshold. Low intensity PEMFs have improved smell in this situation. | Sandyk, Apr 1999 |
Freezing, arrest of speech or handwriting | Difficulty in initiation and smooth processing of repetitive movements. Usually in PD of long duration and advanced stage and is a major cause of disability often resulting in falling. Seen as sudden attack of immobility usually experienced during walking, attempts to turn while walking, or while approaching a destination. For resistance to medication, sometimes a reduction or increase in dose may improve this. Brief low intensity PEMFs improve freezing. The effect of each PEMF treatment lasted several days before appearing again. Weekly PEMFs to the head reduced freezing by ~ 50% and falling by ~ 80-90%, in a six-month follow-up period. | Sandyk, 1996 |
Disorder of body image | A part or parts of bodies perceived as disproportionately large. Low intensity PEMFs have reversed this.
| Sandyk, Feb 1998 |
Speech impairments | More than 89% of PDs. Speech impairments, including severe stuttering, that responded only marginally to medication, improved dramatically. when PEMF treatments given weekly over four years. The speech impairment reappeared when regular PEMF treatments missed. | Sandyk Nov 1997 |
Cognitive impairment | Assessed by drawing tasks, eg drawing a picture of oneself, a bicycle, or a clock face. Drawing impairments indicative of dysfunction of half of the brain. PEMFs of 5 and 7 Hz for 20 minute periods dramatically improve drawing performance, even as soon as after 2 treatments. Drawing performance continues to improve as PEMF treatments continue. | Sandyk, SEPT 1997 |
Very Low intensity PEMFs
Dopamine is considered the most important part of the causes of Parkinson’s disease and is the primary target of medical therapies. Yawning and stretching are dopamine behaviors. When these behaviors are seen in people with neurodegenerative disorders, it is likely that they indicate release of dopamine in the brain. Extremely low intensity PEMFs have been found to increase yawning and stretching.
Various groups also looked at the use of low intensity 8 picoTesla PEMFs in the treatment of Parkinson’s disease, at 2 Hz or 8 Hz, for thirty minutes every forty-eight hours for sixty days. The treatment effects last beyond the time of stimulation depending on symptoms:
less than – 24 hrs for motor impairment,
- 48 hrs for activities of daily living, and
More than – 48 hrs for other symptoms
This shows that chronic stimulation is probably necessary to obtain adequate results with Parkinson’s disease (Bardasano).
The work by Sandyk, with the use of very low intensity PEMFs) also showed that longer-term treatments are necessary to achieve more sustainable results.
Medium intensity transcranial PEMFs
One study looked at a 25 Hz PEMF with intensity of 10 mT (100 gauss) applied for 20 minutes with 10-12 exposures as part of comprehensive rehab therapy in Parkinson’s disease patients. Walking improved significantly and so did the ability to change position. Muscle tension in the lower extremities was reduced in 85% of the patients, vertebral complaints in 85%, and general improvement was reportedly improved in 96% (Jerabek).
In another double-blind clinical study, 97 participants (Hoehn & Yahr stages I-IV) receiving optimal medical anti-Parkinson treatment, were randomized to either active or placebo treatment. Treatment was with transcranial PEMFs daily for a 30-minute home treatment for eight consecutive weeks. The PEMF was 5-8 mT using seven coils over the forehead, two on each side of the head and one in the back of the head. The Parkinson’s Disease Questionnaire (PDQ) was used at baseline and at the end. The active group improved in mobility and activities of daily living (ADL), compared to the placebo group. (Morberg, 2018)
The same group used the same PEMF and protocol on 92 study participants. (Morberg, 2017) Those receiving active treatment had an average age of 67, mean Hoehn & Yahr stage of 2.4, and disease duration of 6 years. In this study the Unified Parkinson’s Disease Rating Scale (UPDRS) was used. There were essentially no differences between the groups. The UPDRS scale is subjective and relatively insensitive and does not exclude the possibility of positive objective physiologic effects, compared to the PDQ.
High intensity PEMFs – repetitive Transcranial Magnetic Stimulation (rTMS)
High intensity PEMFs have been in use to stimulate the brain since 2008 after FDA approval. One type is called repetitive Transcranial Magnetic Stimulation (rTMS). In rTMS, a wire coil is used to generate a magnetic field that can pass through the scalp and the skull to change the excitability in the cortex according to the frequency. High-frequency rTMS (≥5 Hz) – a more rapid pulse rate (pulses per second pr PPS for short) ) induces more excitability in the cortex, while low-frequency/pulse rate rTMS (≤1 Hz) induces an inhibitory effect. A longer duration of stimulation is likely to induce a longer duration of effect. Additionally, there are numerous choices of stimulation sites of rTMS intervention; these sites include the primary motor cortex (M1), which is used mostly for motor symptoms; the dorsolateral prefrontal cortex (DLPFC) is used mostly for depression; the supplementary motor area (SMA) for motor symptoms; and, the cerebellum. The SMA is involved in a variety of cognitive and motor-related processes, in particular, the function of complex chains of movement. (Schramm). rTMS to the cerebellum showed an improvement in stroke (spasticity, balance, and gait), cervical dystonia, Parkinson’s disease (tremor), cerebellar ataxia, and essential tremor but not in multiple sclerosis. (Xia)
Astrocytes (start shaped cells) are specialized brain cells, outnumbering neurons by over five times. Astrocytes are also known collectively as astroglia, and are part of the glial cells in the brain and spinal cord. (Sofroniew) So, the terms astroglia and astrocytes are often used interchangeably. They “tile” the entire central nervous system (CNS) and exert many essential complex functions in the healthy CNS. Brain astrocytes respond to damage and disease in the central nervous system (CNS) through a complex, multifaceted process, called astrogliosis. Dysfunctions of astrocytes (astrogliosis) can lead to or contribute to a variety of CNS disorders. Astroglia/astrocytes undergo significant structural changes in response to CNS damage and disease.
Reactive astrogliosis and exaggerated pro-inflammatory reactions are key pathologic processes in Parkinson’s disease. rTMS has been shown to alleviate neuroinflammation. Higher frequency/pulse rate stimulation of rat brains daily for four weeks had a stronger anti-inflammatory and neuroprotective effect. The Endocannabinoid system (ECS) is integral to neural protection. PEMFs have been found to stimulate the ECS. Actions on the ECS by rTMS are a likely major mechanism of neural protection and anti-inflammatory action positively impacting reactive astrogliosis. (Aceves-Serrano)
High intensity PEMFs like rTMS improve the movement problems of Parkinson’s disease by about 54% overall. Results are better when high pulse rates greater than 5 pulses per second (pps) are used to the motor cortex, for 23% improvement. Results were better for lower than 1 pps applied to the front parts of the brain, with about 50% improvement. A greater number of pulses per session or across sessions have larger benefits. Given the limitations of rTMS treatments, that is, requiring a professional setting with a limited number of treatment options, it appears that treating for more than a week is not much better than treating for less than a week. But this appears to be driven by the number of pulses delivered (Chou).
One rTMS study looked at the treatment of 49 Parkinson’s disease patients. Their symptoms had been well controlled for three months with medications. Patients were divided into four groups to receive various protocols of rTMS with stimulation:
(1) once a day at 3,450 gauss;
(2) twice a day at 3,450 gauss;
(3) twice a day at 5,750 gauss;
(4) twice a day at 8,050 gauss.
Treatments took place for 10 days. The patients were evaluated 3 times before rTMS to obtain baseline data, then on days 3 and 7 of treatment, and then at 1 and 3 months after the end of treatment. They measured range of movement and disability in activities of daily living and short-term memory. In group 1, treatment once per day, there were no significant changes in symptoms at any time. By contrast, all other groups (receiving treatment twice per day) had significant improvement in symptoms at the 1-month assessment. But there were no differences in scores between the treated groups. At the 3-month evaluation point, the Parkinson’s disease symptom scores were still significantly better than baseline values but there were significant inter-group differences, with group 3 (twice a day at 5,750 gauss) showing the most improvement. The results show that this protocol causes long-lasting dose-related symptom improvements in Parkinson’s disease and may even allow dose reduction in medication (Mally).
In another rTMS study, Parkinson’s disease patients were evaluated to see if rTMS could improve muscle function assessed by simple reaction times. It had been previously shown that simple reaction time was significantly improved in normal individuals and in those with Parkinson’s disease. Assessments were done at the time of peak medication effect. For comparison, the same tasks were studied in ten normal volunteers. There were significant differences between Parkinson’s disease patients and normal volunteers during rTMS with the coil on the head. In Parkinson’s disease patients, rTMS significantly shortened reaction time and movement time without affecting errors. The mean performance of Parkinson’s disease patients improved during rTMS and patients reported that it was easier to perform the test during rTMS. All patients were significantly slower in the unmedicated state compared with the medicated state. Their performance in the unmedicated state improved significantly with PEMF stimulation (Pascual-Leone).
In a separate analysis of multiple studies, they evaluated the possible value of rTMS on cognitive function in Parkinson’s disease. They evaluated 12 studies and found that there was a mild short-term effect of rTMS on global cognition, executive function, attention and working memory. There were no significant benefits for long-term outcomes. The authors noted that more research is needed with larger numbers of individuals being tested. (He)
There is a frequently expressed concern about using high intensity PEMFs to the brain. Because some studies have suggested that an increase in stimulation pulse rate might enhance therapeutic efficacy, a randomized controlled trial was conducted (Benninger) using 50 pps/Hz rTMS in 26 individuals with mild to moderate Parkinson’s disease. Stimulation was to the motor cortex in 8 sessions over 2 weeks. Testing included evaluation of gait, slow movement, UPDRS and additional clinical, neurophysiological and neuropsychological elements. In addition, they studied the safety of our TMS with electromyography – electroencephalogram (EMG – EEG) monitoring during and after stimulation. The 50 pps rTMS did not improve gait, slow movement and global and motor UPDRS measures. There was a short-lived improvement in ADL. The neurophysiological and neuropsychological measures did not change. EMG/EEG showed no pathological increase of brain action or epileptic risk. There were no adverse effects. So, while this study did not find any clear benefits in movement and functional status, it did reveal the safety, even in those with Parkinson’s disease. Unfortunately, this research focused on stimulation of the motor cortex and not on the source lesions of the Parkinson’s disease in the substantia nigra/subthalamic nucleus. So, at this point it is not known whether stimulating the damaged tissue directly would produce any better benefits.
Parkinson’s disease has traditionally been considered a single-region disease in the basal ganglia but has recently been proposed as a system-level disorder that includes functional interactions between brain regions such as the dopamine dependent motor circuit (cortico-striatal-thalamic circuit) and the cognitive circuit (frontal-striatal circuit). However, structural changes in the brain take time to be produced and eight weeks of treatment is unlikely to be sufficient. Thus, even longer treatment periods are needed to better understand the potential of this treatment modality (Jensen)
Physiologically, it is known that erythropoietin (EPO), vascular endothelial growth factor (VEGF), as well as dopamine are present in the brain. EPO has neuroprotective benefits and neuro repair as seen in animal studies. In spinal cord traumatic injury and Alzheimer’s animal research, EPO improved memory and spatial learning and had a neuroprotective effect. VEGF affects growth of new blood vessels (angiogenesis), neural migration and neuroprotection and, along with EPO has the potential to help neurological disorders.
Since it was speculated that PEMFs could enhance neuroprotection, research was done to test this theory that long-term treatment with transcranial PEMF (T-PEMF) would improve motor performance (in terms of movement speed) and stimulate production of neuroprotective and angiogenetic compounds (EPO and VEGF) in the brain in patients with Parkinson’s disease. (Jensen) Using the same protocol as Morberg, the authors tested the idea that long-term treatment with T-PEMF would improve motor performance (in terms of movement speed) and stimulate production of neuroprotective and angiogenetic compounds (EPO and VEGF) in the brain in patients with Parkinson’s disease.
The active T-PEMF treatment group had 16 patients and 8 were included in the Parkinson’s disease-control no T-PEMF treatment group. Disease severity was assessed using the UPDRS. Daily 30 min home T-PEMF of 5-8 mT was applied for three eight-week periods, separated by one-week pauses. The total treatment period was 26 weeks. The primary outcome was movement speed, which was assessed in a timed six-cycle sit-to-stand (STS) task, where the participants were asked to perform the task as fast as possible. Examinations were done at week 17 and week 27. Eight patients from the T-PEMF group had lumbar puncture just before treatment and within one day after treatment completion for their brain EPO and VEGF levels. Results of the STS test improved progressively over the 26 weeks by about 20%. At the previously studied (Morberg, 2017) 8 wk study point it improved only by about 8%. The graph below (taken from Jensen), shows that 26 weeks of treatment improved STS (green line) from 100% at baseline (0%) to 80%. The control group (redline) got worse over that time by about 2%.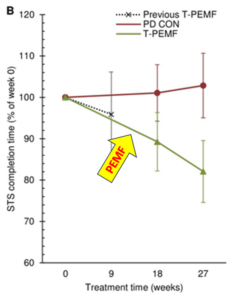
Spinal fluid (CSF) EPO concentrations increased significantly in response to the T-PEMF treatment intervention (all patients increased (n = 8)). CSF-VEGF concentration increased in five out of six patients. So, it appears that T-PEMF treatment has contributed to neural repair and protection of the dopamine neurons in this study. In addition, importantly, there was no difference between the longer treatment time group and the placebo group regarding reported adverse events.
The previous study (Morberg, 2017) showed that an eight-week treatment period was beneficial for patients with mild Parkinson’s disease whereas the treatment effect was less among the more severely affected participants. However, the present study (Jensen) treatment for 30 min/day for 3×8 weeks showed a benefit for the entire study group including mild as well as more severely affected Parkinson’s disease patients. It’s also plausible that the longer treatment time would have impacted the amount of EPO and VEGF more as well. Finally, even though the treatment group was well medicated, T-PEMF treatment still improved the completion time in the STS-task by 19%, comparably to the healthy control group. This shows that the PEMF treatment improves significantly on the benefits seen from medication management. The question then becomes whether medication management is necessary when doing more intensive longer-term PEMF therapy. However, this difference in benefit may only apply to this study and to the motor function studied, and may not apply to the other aspects of dysfunction in Parkinson’s disease.
Freezing of Gait (FOG): The effect of rTMS has been studied on FOG. However, stimulation of neither the motor cortex nor the frontal cortex showed benefit in one study. (Kim KW). Another stimulation site, The supplementary motor area (SMA) has also been explored. When tested on 30 Parkinson’s disease patients with FOG, it was shown that 10 sessions of 10 pps rTMS over the SMA improved FOG (Mi). This study also found that the benefit could last at least 4 weeks after stimulation. This result was consistent with those of another study (Kim SJ), where they found significant improvements after 2 sessions of high-PPS SMA stimulation in 12 Parkinson’s disease patients, but not after motor cortex stimulation. These results suggested that SMA stimulation may be a more appropriate target in Parkinson’s disease patients with FOG.
How to use PEMFs for the treatment of Parkinson’s disease
PEMFs can be extraordinary helpful in the care of Parkinson’s disease and are even more valuable considering that no other therapies to date have been able to cure it or stop its progression. Even the standard medication, the primary line of treatment for Parkinson’s disease, is only about 50% effective overall to help with the symptoms of Parkinson’s disease. While research is ongoing to develop new therapies and approaches, PEMF therapy has a long history of helping a myriad of different health conditions, safely and without risk. In fact, when somebody owns a PEMF device, not only do they benefit across many different health needs, but it can also be used by other family or household members, including pets. There are many scientifically backed examples of the extent of usefulness of PEMFs in the book “Power Tools for Health” (Pawluk), including a section on its use for Parkinson’s disease.
As mentioned earlier, even twice-weekly treatments twice-weekly treatments with extremely low intensity PEMFs applied to the head for ten weeks has been shown to eliminate the daily drop-off of medication benefit. (Sandyk, Oct 1997) At ten weeks after starting the PEMFs, there was 40% improvement in response to medication with minimum change in efficacy during the course of the day or evening. They have even been found to be helpful for the side effects of the medications. In addition, since decline in the response to medication over time in patients with Parkinson’s disease is associated with progression of the disease, even intermittent application of PEMFs may reverse the course of progressive Parkinson’s disease.
Those results are based on a health services delivery model requiring that the treatment be provided in a doctor’s office. This is not only inconvenient and also expensive over time, but also increases the risk of progressive disease. As Parkinson’s disease progresses, becomes much more difficult to manage, and as a result there is a much greater toll on the health, function and vitality of the person.
Overall health
Because of all the different actions of PEMFs, reviewed in “Power Tools for Health“, PEMFs are not just a treatment for Parkinson’s disease but also overall health. Any other health demands place a significant strain on the body in coping with Parkinson’s disease, and increase the risk of more rapid progression. As a result, PEMFs not only help with Parkinson’s disease but any other health conditions (comorbidities) as well. Since Parkinson’s disease typically begins after age 50, and especially after age 60, comorbid conditions are common. Per the CDC many people have have 2 or more comorbid conditions: 33% of those between 45-64 years of age and 64% of those 65 or older. (Boersma)
Progression of PEMF benefits
As PEMFs are used there is a progression of benefits, dependent on the amount of healing that happens with use. Often there are significant improvements in symptoms in the first few days to weeks. The extent of these improvements may depend on the pace of increasing treatment time and intensity, that is, the “going low and slow” progression. Clearly, the lower the intensities used and the less the treatment time, the less the expected benefit. While anybody can get benefit even at the lower intensities and time, the better benefits will happen with more treatment time and higher magnetic field intensities.
After the initial “blush” of changes in symptoms, with continuing treatment there will be gradual improvements in function, either improved function over normal or less functional disability. As healing continues, the improvements in symptoms and function last longer and are less likely to return as quickly. There may come a time when there appear to be no further improvements in symptoms and function. People often worry that this means that “tolerance” to the magnetic field therapy is happening. This just means that the “low hanging fruit” of the benefits of PEMFs have been achieved. At this point continued therapy will be working at the deeper levels of healing, at the cellular level. Cellular level healing tends to take longer, especially with the brain, and is much less obvious and noticeable. I can generally say that “cells don’t talk to you” but tissues and organs do. When enough of the deeper cellular healing accumulates, the improvements become more noticeable and are less likely to regress when treatment is reduced. If treatment is reduced too soon, symptoms are more likely to recur. Generally, the longer it takes symptoms to recur after stopping or reducing treatment, the deeper the healing. But if symptoms do recur that means that healing is not completed. It’s possible that some levels of damage in the body can never be completely fixed and we may have to accept the level of gain obtained to that point.
Let’s not forget that Parkinson’s disease is not just a brain disorder. It is a systemic disorder that has brain effects as a major manifestation. Most of the things that happen to the brain and nervous system from the PEMFs are also happening in the rest of the body. In Parkinson’s disease jargon, there are the motor benefits and the nonmotor benefits. PEMFs help with both groups of needs. The order in which they are helped is controlled by the body and we can prioritize which ones are going to be helped first, and so forth.
In the treatment of Parkinson’s disease, the actions of PEMFs that are most important (Pawluk) include reductions in:

Magnetic field intensity matters
The intensity of the magnetic field is very important in being able to reach deep into the brain with sufficient magnetic field intensity to reduce inflammation and stimulate many of the other actons of PEMFs. Our experience is that the benefits of very low intensity PEMFs that have been published are typically not long-lasting and not as beneficial as higher intensity PEMFs, as can be seen from the research on rTMS. The challenge in showing long-term effectiveness of rTMS is because of the nature of rTMS research. It usually has not been done long enough to see sustainable and significant benefits.
It is recommended to read about the role of adenosine in reducing inflammation in the body and that the optimal magnetic field intensity for the best impact on inflammation. More can be read about this at https://www.drpawluk.com/pain-inflammation-adenosine/. Also, it is strongly recommended to read the books https://www.drpawluk.com/product/power-tools-for-health/ and https://www.drpawluk.com/product/supercharge-your-health-with-pemf-therapy/ for more in-depth information on PEMF therapies. The “Power Tools for Health” book has over 500 references for those who wish more of the scientific background. The “Supercharge Your Health” gives much more practical advice on the use of PEMFs, in general and more specifically for about 80 different health conditions.
Further advice
Given the progression of Parkinson’s disease over time and the irreversibility of the physical changes with later stages of Parkinson’s disease, it is strongly recommended to start as early as possible, certainly at the time of initial diagnosis. As screening tests become more available it may be possible to start treatment for prevention purposes very early, before motor symptoms become obvious. Tests are becoming available now for detecting early levels of alpha-synuclein either with intestinal biopsies or spinal fluid. Inflammation is a major driver of causing aggregation of the normal tissue levels of synuclein. As the research is showing, once the aggregated synuclein begins, it starts to migrate, eventually ending up in the nervous system. One of the key drivers of the aggregation of synuclein is inflammation, whether in the gut or elsewhere in the body. Therefore, starting as early as possible in the course of the disease is essential to limit its progression and limits the course of the disease.
Because of its chronic local and systemic nature, lifetime daily home use of a PEMF system with adequate intensity is recommended before irreversible damage happens in the body, and especially in the brain. The minimum recommended intensity is about 4000 Gauss, to be able to reach deep into the abdomen and across the brain. Treatment to the brain should be at the top of the neck with the applicators placed from the back of the head toward the front of the brain. Additional placements could be of a double loop coils over the top of the head, with a loop over each ear. If there are significant cognitive, memory and mood issues, placements could also be across the front of the head. Ultimately, each individual will have to use trial and error to determine the best placements.
In addition to local treatments to the brain, daily or even twice daily whole-body treatment should also be used.
PEMF therapies can be combined readily with almost any other kinds of therapies, and should usually include neuro-supportive nutrition and supplements. PEMFs can also be combined with almost any other kinds of complementary therapies, although many of these do not work deep enough. These other therapies can include red light, intravenous nutrients, such as IV vitamin C, glutathione, and alpha lipoic acid (ALA). It is advised to seek the support of a clinician/professional who is familiar with recommending supplements for neurological disorders. Some of this information about combining PEMFs with other therapies is found in the two books mentioned above.
Resources
https://www.drpawluk.com
https://www.drpawluk.com/pain-inflammation-adenosine/.
https://www.drpawluk.com/product/power-tools-for-health/ https://www.drpawluk.com/product/supercharge-your-health-with-pemf-therapy/
PEMF consultations
To be certain to obtain the right PEMF system and be trained in its proper use, it is recommended to seek the support of a licensed professional who is familiar with PEMF therapy and other complementary therapies to go with it. It PEMF therapies are recommended, always ask about the intensity of the PEMF system being used. Remember, that office-based treatments are not going to be done affordably over extended periods of time to be certain that the best results will be obtained, especially considering that treatment should be lifetime. As yet, there is no cure still, even with PEMF therapy, so, basically, lifetime maintenance and control is necessary as far as we know.
Consultations without charge are available on Dr. Pawluk.com. https://www.drpawluk.com/consult/
Summary
So, it appears that PEMFs of various intensities and treatment times can be helpful in the management of Parkinson’s disease. Longer treatment times and stronger PEMFs are very much likely to produce better and more lasting results. Since the goal is to help to repair the brain, not just improve function temporarily, long-term treatment frequently appears to be necessary. What is not known is whether the combination of periodic high intensity PEMFs along with a home therapy program using a lower intensity PEMF system may be the most helpful. It also appears that PEMFs may be synergistic with medication, which has a history of losing its effectiveness over time. It is not known how well PEMFs alone, without medication, may be able to help the symptoms and progression of Parkinson’s disease. At this point at least, there’s still no clear evidence that PEMFs can “cure” the condition. Much of the research with rTMS follows typical protocols, either to the motor area of the brain or the left frontal lobe. I have not seen any research that focuses treatment directly over the anatomic area in the brain, that is, the substantia nigra, primarily involved in the condition. Whole body treatment is recommended, in addition to the brain, because it appears that the origins of Parkinson’s disease may well be elsewhere in the body, especially in the intestinal tract.
References
- Aceves-Serrano L, Neva JL, Doudet DJ. Insight Into the Effects of Clinical Repetitive Transcranial Magnetic Stimulation on the Brain From Positron Emission Tomography and Magnetic Resonance Imaging Studies: A Narrative Review. Front Neurosci. 2022 Feb 21;16:787403.
- Benninger DH, Iseki K, Kranick S, et al. Controlled study of 50-Hz repetitive transcranial magnetic stimulation for the treatment of Parkinson disease. Neurorehabil Neural Repair. 2012 Nov-Dec;26(9):1096-105.
- Boersma P, Black LI, Ward BW. Prevalence of Multiple Chronic Conditions Among US Adults, 2018. Prev Chronic Dis 2020;17:200130.
- Chou YH, Hickey PT, Sundman M, et al. Effects of repetitive transcranial magnetic stimulation on motor symptoms in Parkinson disease: a systematic review and meta-analysis. JAMA Neurol. 2015 Apr;72(4):432-40.
- He PK, Wang LM, Chen JN, et al. Repetitive transcranial magnetic stimulation (rTMS) fails to improve cognition in patients with Parkinson’s disease: a Meta-analysis of randomized controlled trials. Int J Neurosci. 2022 Mar;132(3):269-282.
- Jensen BR, Malling ASB, Schmidt SI, et al. (2021) Long term treatment with transcranial pulsed electromagnetic fields improves movement speed and elevates cerebrospinal erythropoietin in Parkinson’s disease. PLoS ONE 16(4): e0248800.
- Jerabek, J and Pawluk, W. Magnetic therapy in Eastern Europe: a review of 30 years of research. Publ. Advanced Magnetic Research of the Delaware Valley, Chicago, 1996.
- Kim SJ, Paeng SH, Kang SY. Stimulation in supplementary motor area versus motor cortex for freezing of gait in Parkinson’s disease. J Clin Neurol. 2018;14(3):320–326.
- Kim YW, Shin IS, Moon HI, Lee SC, Yoon SY. Effects of non-invasive brain stimulation on freezing of gait in Parkinsonism: a systematic review with meta-analysis. Parkinsonism Relat Disord. 2019;64:82–89.
- Mally J and Stone TW. Therapeutic and “dose-dependent” effect of repetitive microelectroshock-induced by transcranial magnetic stimulation in Parkinson’s Disease. J Neurosci Res 57(6):935-940, 1999.
- Morberg BM, Malling AS, Jensen BR, et al. Parkinson’s disease and transcranial pulsed electromagnetic fields: A randomized clinical trial. Mov Disord. 2017 Apr;32(4):625-626.
- Morberg BM, Malling AS, Jensen BR, et al. The Effects of Transcranial Pulsed Electromagnetic Field stimulation on quality of life in Parkinson’s Disease. Eur J Neurol. 2018 Mar 23.
- Pascual-Leone A, Valls-Sole J, Brasil-Neto JP, et al. Akinesia in Parkinson’s disease. ii. Effects of subthreshold repetitive transcranial motor cortex stimulation. Neurology 44(5):892-898, 1994.
- Pawluk W and Layne CJ. Power Tools for Health: how magnetic fields (PEMFs) help you. Publ. Friesen Press, 2017.
- Sandyk R. Freezing of gait in Parkinson’s disease is improved by treatment with weak electromagnetic fields. Int J Neurosci. Mar;85(1-2):111-24, 1996.
- Sandyk R. Reversal of a body image disorder (macrosomatognosia) in Parkinson’s disease by treatment with AC pulsed electromagnetic fields. Int J Neurosci. Feb;93(1-2):43-54, 1998.
- Sandyk R. Reversal of cognitive impairment in an elderly parkinsonian patient by transcranial application of picotesla electromagnetic fields. Int J Neurosci. 1997 Sep;91(1-2):57-68.
- Sandyk R. Speech impairment in Parkinson’s disease is improved by transcranial application of electromagnetic fields. Int J Neurosci. Nov;92(1-2):63-72, 1997.
- Sandyk R. Treatment with AC pulsed electromagnetic fields improves olfactory function in Parkinson’s disease. Int J Neurosci. Apr;97(3-4):225-33, 1999.
- Sandyk R. Treatment with AC pulsed electromagnetic fields improves the response to levodopa in Parkinson’s disease. Int J Neurosci. Oct;91(3-4):189-97, 1997.
- Sandyk R. Yawning and stretching induced by transcranial application of AC pulsed electromagnetic fields in Parkinson’s disease. Int J Neurosci. Mar;97(1-2):139-45, 1999.
- Schramm, S., Albers, L., Ille, S. et al. Navigated transcranial magnetic stimulation of the supplementary motor cortex disrupts fine motor skills in healthy adults. Sci Rep 9, 17744 (2019).
- Sofroniew MV. Astrogliosis. Cold Spring Harb Perspect Biol. 2014 Nov 7;7(2):a020420.
- Xia Y, Wang M, Zhu Y. The Effect of Cerebellar rTMS on Modulating Motor Dysfunction in Neurological Disorders: a Systematic Review. Cerebellum. 2023 Oct;22(5):954-972. Parkinson’s disease