Joint Replacements And Implanted Prosthetics
Table of Contents
IMPROVEMENTS IN JOINT REPLACEMENT PROCEDURES
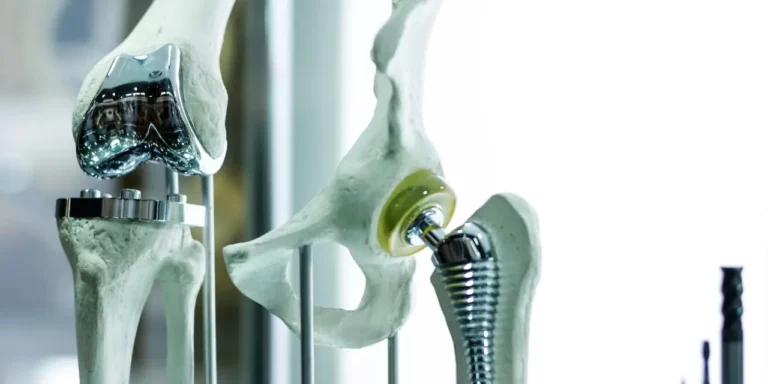
The last 20-30 years have witnessed a marked increase in total joint replacement procedures with excellent results. Total or partial hip and knee replacements are the most common. There are more than 1 million Americans having one of these procedures each year. The main complication is aseptic (noninfectious) loosening. It is the cause of more than 70% of hip revisions and more than 40% of knee revisions.
Cementless hip replacement will fail in as many as 25% of young patients after 10 years. Significant bone loss is seen in up to 14% of individuals during the first 3 months after an initial total hip replacement. Pain is a common finding after loosening occurs, on average becoming a problem after 12 months.
Revision prostheses have poorer outcomes compared with primary joint replacement. This is because the quality of the bone tissue where the new prosthesis is to be implanted is poor. It has loss of bone mass and osteoporosis of the surrounding bone. Furthermore, there is breakdown of bone around the implant, bone loss, and poor natural bone healing capability. These are the main problems in reconstructive hip surgery and reduce the lifespan of revision implants. Inflammation produces enzymes that degrade tissue at the bone implant interface.
PEMF therapy has been shown to improve bone mass, decrease harmful inflammation, and stimulate circulation. Therefore, it has been studied in conjunction with joint replacements with much success.
A PEMF STUDY ON HIP REVISION PATIENTS
In one randomized double-blind study, 30 patients undergoing hip revision were treated for 6 hours per day for 90 days after surgery. Subjective improvement was higher in those receiving the PEMF treatments compared to the placebo. Patients whose bone density measurements (DXA) improved more than 3.5% were considered responders. Various “bone zones” were tested; some results were the same in the PEMF and placebo groups. In two of the zones (corresponding to the inside of the bone lining), researchers found that 40% of patients were responders in the control/placebo group, compared to up to between 66% and 93% responders in the PEMF group. This study shows a positive clinical correlation between PEMF therapy and bone stock restoration after surgery.
Researchers have studied a variety of new approaches to improving the bone-implant interface. Some of these include other types of implant surfaces, locally-applied osteoporosis medications (bisphosphonates), and locally-applied growth factors, platelet-rich plasma, and stem cells. However, costs, safety issues, complexity of administration, optimal dosing, and lack of long-term studies limit these options.
Surgeons doing an original joint replacement or implant often encourage the patient to wait until they are older to get the procedure done because of the very real possibility that in 10 to 15 years the procedure will have to be redone. Redone procedures are more complex and challenging, and have an increased risk of breakdown.
New, noninvasive strategies are needed to enhance the success of the implantation procedure by increasing bone formation around the prosthesis and lowering local inflammation, especially in cementless implants. PEMF stimulation is ideal for this. The effectiveness of PEMFs in enhancing endogenous bone repair and reducing inflammatory processes has been shown in multiple studies.
PEMFS CAN SUPPORT THE BONE AND THE MATERIALS USED TO HELP THE BONE
PEMFs have been studied for the ability of various materials to integrate with bone. Almost all the studies of shown that PEMF stimulation can be applied locally and can significantly enhance the integration of implant materials, including titanium, stainless steel and ceramic implants. Even nails or rods implanted into the bone marrow of long bones, such as the femur, which were movable or unstable, as would happen with surgery for fractures, improved and were integrated into the bone better.
I had an experience myself with a titanium dental implant, where the implant was integrated better into a bone graft because I was using PEMF stimulation. Even the dental surgeon was pleased, surprised, and amazed. He had apparently only seen this type of integration one other time in his 17 years of practice.
PEMFs have been studied for the osteointegration of joint replacement prostheses. There are 2 aspects to this: the treatment of loosened prostheses and the use of PEMFs after a revision.
MORE PEMF STUDIES SUPPORT BONE HEALTH
In the first scenario (treatment of loosened prostheses) the intention is to reduce the need for a revision. In one study, 132 patients had PEMF therapy for advanced loosening of their prosthesis. Treatment was done 2-3 times a day for 40 minutes each time, for 20 weeks. Follow up was done over the course of 5 years. A revision procedure was no longer deemed necessary in 70% of patients.
In an extension of this research, PEMF therapy was administered to more than 1,000 patients with loosened artificial hips. The PEMF signal used was 30 gauss with frequencies ranging from 2 to 20 Hz. The treatment lasted for either 6 months, or until patients reported complete relief from pain and discomfort, whichever came first. Treatment was successful in 70% of the patients. Before treatment, 76% used crutches; this was reduced to 48% after the study. In more than 65% of the patients, further surgery could be avoided within a follow-up of 10 years. The treatment took an average of 16 weeks. Before treatment, 54% of the patients suffered from permanent pain; this was reduced down to 6.5% afterwards. Before PEMF treatment, 36% of patients used analgesics and after treatment only 2% did. Researchers concluded that PEMFs are best considered for patients at an early stage of aseptic loosening.
In another double-blind study using PEMFs for loosened cement hip prostheses, 37 patients completed 6 months of treatment (either active or placebo). Success was determined clinically using a Harris hip score greater than or equal to 80 points. Ten of the 19 active patients (53%) were considered successes, compared to two of the 18 placebo patients. This is a statistically significant and clinically relevant result. A 60% relapse rate among the active successes was seen at 14 months after stimulation, and despite maintenance therapy of one hour per day, the relapse rate increased to 90% at three years. These data suggest that for loosened cemented hip prostheses, use of PEMFs is a treatment option only to delay revision hip surgery.
Loosening in the absence of infection (aseptic) is the most common problem of hip replacements, limiting their long-term success. There was a study of PEMF treatment in 24 patients with this complication. At the end of treatment, six months and one year later, pain and hip movements improved significantly. Both bone scans and ultrasonography improved significantly, but not in plain X-ray. The decreased pain and improved function suggest that PEMF is effective in improving symptoms of patients with loose hip replacement, supported by objective improvements in bone scan and ultrasound. No improvement, however, can be expected in patients with severe pain due to gross loosening.
Another group of 30 patients undergoing hip revision with a replacement prosthesis were treated with a 20 gauss PEMF signal for 6 hours per day, starting from the 7th through 90th days after revision in a double-blind study. PEMF-treated individuals were functionally better. Postoperative bone mineral density (BMD) was 66–93% versus 40% in controls, or more than double the improvement, even at 90 days after surgery. In addition, the PEMF group had a reduction in pain of 77% compared to 40% in the control, even as far out as 90 days after the procedure. The treatment was not associated with any negative side effects; nevertheless, it must be noted that the use of the electromagnetic stimulation at the hip required considerable patient commitment. Still, this important study showed that PEMF treatment aids clinical recovery and bone restoration.
In another study, 45 patients were studied using a 75 Hz, 20 gauss PEMF stimulator for 60 days, at a minimum of 6 hours per day. Of those, 76% had good or excellent results. The more treatment that was done, the better the results were, with 80% of those who used it for more than 30 days reporting good results. But, of those who used it for more than 60 days with at least 360 hours of exposure, 92% had good results. There appears to be a dose-related effect which is possibly cumulative. No side effects of stimulation were seen.
In addition to the benefits seen with PEMFs in the treatment of loosened implants, the therapy has also been studied immediately following joint replacement surgery, with the long-term goal being extended life of the implant and prevention of loosening in the first place.
During the healing process, bone cells first proliferate, then mature, and finally deposit minerals. In the active growth phase, osteoblasts have elevated production of extracellular matrix (ECM) genes such as type I collagen (COL I). When cells enter the maturation phase, cell growth slows down and the expression of matrix formation proteins such as COL I and alkaline phosphatase (ALP) increase. The last stage involves adding minerals to the area of the injured soft tissue. Since inflammation can hinder bone repair, it is important to know whether PEMF could stimulate bone repair under conditions of inflammation. Bone implants themselves lead to inflammation, which can hinder the progress of bone repair.
Conditions of bone repair were studied in experiments simulating implant placement. On day 7, the PEMF-exposed bone culture released more nitric oxide (NO) than the control. PEMFs resulted in a significant increase in NO release. PEMF-induced NO production in macrophages takes on an oscillating pattern and peaks at 7 days. The survival of osteoblasts in a control group decreased from days 0 to 7. The PEMF-exposed osteoblasts had significantly higher survival on day 7. Osteoblasts stimulated by PEMF began to synthesize internal NO and probably developed their own protective mechanisms such as intracellular detoxifying agents and heat-shock proteins to prevent NO from damaging themselves. NO subsequently promoted osteoblastic activities such as growth, viability and collagen expression.
As a result of increased collagen synthesis in the ECM, the cells produced elevated alkaline phosphatase (ALP) activity. Higher ALP activity eventually leads to more mineral deposition and superior bone repair. The high osteoblast proliferation stimulated by PEMF is the primary determinant of the rate of bone formation.
The above studies show a strong correlation between PEMF therapy and successful treatment and longevity of joint replacement implants. There appears to be a dosing effect where longer treatment times or treatments at higher intensities have higher long-term success than shorter treatment times or lower intensity treatments.